
Back أدرافينيل Arabic ادرافینیل AZB Adrafinil German Adrafinilo Spanish Adrafiniili Finnish Adrafinil French Adrafinil Galician Adrafinil Italian Adrafinil Dutch Adrafinil Polish
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Olmifon |
| Other names | CRL-40028 |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 80% |
| Metabolism | 75% (liver) |
| Metabolites | Modafinil |
| Elimination half-life | 1 hour (T1/2 is 12–15 hours for modafinil)[5] |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.058.440 |
| Chemical and physical data | |
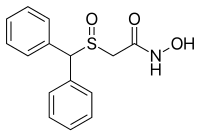
| Formula | C15H15NO3S |
| Molar mass | 289.35 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Adrafinil, sold under the brand name Olmifon, is a wakefulness-promoting medication that was formerly used in France to improve alertness, attention, wakefulness, and mood, particularly in the elderly.[6][7][8] It was also used off-label by individuals who wished to avoid fatigue, such as night workers or others who needed to stay awake and alert for long periods of time. Additionally, the medication has been used non-medically as a novel vigilance-promoting agent.[6]
Adrafinil is a prodrug; it is primarily metabolized in vivo to modafinil, resulting in very similar pharmacological effects.[6] Unlike modafinil, however, it takes time for the metabolite to accumulate to active levels in the bloodstream. Effects usually are apparent within 45–60 minutes when taken orally on an empty stomach.[citation needed]
Adrafinil was marketed in France until September 2011 when it was voluntarily discontinued due to an unfavorable risk–benefit ratio.[7]
- ^ "Peak Nootropics LLC aka Advanced Nootropics - 557887 - 02/05/2019". Center for Food Safety and Applied Nutrition (CFSAN). U.S. Food and Drug Administration. 2019-12-20. Retrieved 2023-05-05.
- ^ Office of Regulatory Affairs (2019-12-20). "Hermitage Man Sentenced for Importing and Selling Drugs Not Approved by FDA". U.S. Department of Justice – via U.S. Food and Drug Administration.
- ^ "Fort Collins Couple Sentenced to Federal Prison for Illegally Selling Unapproved Drugs". Food and Drug Administration. FDA Office of Criminal Investigations. June 10, 2022. Retrieved May 21, 2023.
- ^ "Arizona Company and CEO Plead Guilty to the Distribution of Drugs Not Approved by the FDA and Will Pay $2.4 Million". Food and Drug Administration. FDA Office of Criminal Investigations. October 30, 2023. Retrieved November 6, 2023.
- ^ Robertson P, Hellriegel ET (2003). "Clinical pharmacokinetic profile of modafinil". Clin Pharmacokinet. 42 (2): 123–37. doi:10.2165/00003088-200342020-00002. PMID 12537513. S2CID 1266677.
- ^ a b c Milgram N (1999). "Adrafinil: A Novel Vigilance Promoting Agent". CNS Drug Reviews. 5 (3): 193–212. doi:10.1111/j.1527-3458.1999.tb00100.x.
- ^ a b AFSSAPS (2011). "Point d'information sur les dossiers discutés en commission d'AMM Séance du jeudi 1er décembre 2011 - Communiqué". Archived from the original on 13 September 2017.
- ^ Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 20–. ISBN 978-3-88763-075-1.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search