
Back دوبامين (دواء) Arabic Dopamina (medicación) Spanish Dopamina (sendagaia) Basque Դոպամին (դեղորայք) Armenian Допамін (лікарський засіб) Ukrainian Dopamine (thuốc) Vietnamese 多巴胺 (藥物) Chinese
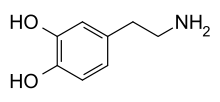 Skeletal formula of dopamine | |
 | |
| Clinical data | |
|---|---|
| Trade names | Intropin, Dopastat, Revimine, others |
| Other names | 2-(3,4-Dihydroxyphenyl)ethylamine; 3,4-Dihydroxyphenethylamine; 3-hydroxytyramine; DA; Intropin; Revivan; Oxytyramine; Prolactin inhibiting factor; Prolactin inhibiting hormone |
| AHFS/Drugs.com | Monograph |
| Routes of administration | Intravenous injection |
| ATC code | |
| Physiological data | |
| Source tissues | Substantia nigra; ventral tegmental area; many others |
| Target tissues | System-wide |
| Receptors | D1, D2, D3, D4, D5, TAAR1[2] |
| Agonists | Direct: apomorphine, bromocriptine Indirect: cocaine, substituted amphetamine, cathinone, bupropion |
| Antagonists | Neuroleptics, metoclopramide, domperidone |
| Metabolism | MAO, COMT,[2] ALDH, DBH, MAO-A, MAO-B, COMT |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | MAO, COMT,[2] ALDH, DBH, MAO-A, MAO-B, COMT |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C8H11NO2 |
| Molar mass | 153.181 g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.26 g/cm3 |
| Melting point | 128 °C (262 °F) |
| Boiling point | decomposes |
| |
| |
| (verify) | |
Dopamine, sold under the brand name Intropin among others, is a medication most commonly used in the treatment of very low blood pressure, a slow heart rate that is causing symptoms, and, if epinephrine is not available, cardiac arrest.[4] In newborn babies it continues to be the preferred treatment for very low blood pressure.[5] In children epinephrine or norepinephrine is generally preferred while in adults norepinephrine is generally preferred for very low blood pressure.[6][7] It is given intravenously or intraosseously as a continuous infusion.[4] Effects typically begin within five minutes.[4] Doses are then increased to effect.[4]
Common side effects include worsening kidney function, an irregular heartbeat, chest pain, vomiting, headache, or anxiety.[4] If it enters into the soft tissue around the vein local tissue death may occur.[4] The medication phentolamine can be given to try to decrease this risk.[4] It is unclear if dopamine is safe to use during pregnancy or breastfeeding.[4] At low doses dopamine mainly triggers dopamine receptors and β1-adrenergic receptors while at high doses it works via α-adrenergic receptors.[4]
Dopamine was first synthesized in a laboratory in 1910 by George Barger and James Ewens in England.[8] It is on the World Health Organization's List of Essential Medicines.[9] In human physiology dopamine is a neurotransmitter as well as a hormone.[10]
- ^ Cruickshank L, Kennedy AR, Shankland N (2013). "Tautomeric and ionisation forms of dopamine and tyramine in the solid state". J. Mol. Struct. 1051: 132–136. Bibcode:2013JMoSt1051..132C. doi:10.1016/j.molstruc.2013.08.002.
- ^ a b "Dopamine: Biological activity". IUPHAR/BPS guide to pharmacology. International Union of Basic and Clinical Pharmacology. Archived from the original on 5 February 2016. Retrieved 29 January 2016.
- ^ Cite error: The named reference
Neoatricon EPARwas invoked but never defined (see the help page). - ^ a b c d e f g h i "Dopamine Hydrochloride". drugs.com. American Society of Health-System Pharmacists. 29 June 2016. Archived from the original on 14 September 2016. Retrieved 15 July 2016.
- ^ Bhayat SI, Gowda HM, Eisenhut M (May 2016). "Should dopamine be the first line inotrope in the treatment of neonatal hypotension? Review of the evidence". World Journal of Clinical Pediatrics. 5 (2): 212–222. doi:10.5409/wjcp.v5.i2.212. PMC 4857235. PMID 27170932.
- ^ De Backer D, Aldecoa C, Njimi H, Vincent JL (March 2012). "Dopamine versus norepinephrine in the treatment of septic shock: a meta-analysis*". Critical Care Medicine. 40 (3): 725–730. doi:10.1097/ccm.0b013e31823778ee. PMID 22036860. S2CID 24620964.
- ^ Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, et al. (February 2013). "Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012". Critical Care Medicine. 41 (2): 580–637. doi:10.1097/CCM.0b013e31827e83af. PMID 23353941. S2CID 34855187.
{{cite journal}}: CS1 maint: overridden setting (link) - ^ Fahn S (2008). "The history of dopamine and levodopa in the treatment of Parkinson's disease". Movement Disorders. 23 (Suppl 3): S497–S508. doi:10.1002/mds.22028. PMID 18781671. S2CID 45572523.
According to Hornykiewicz,6 dopamine was first synthesized by George Barger and James Ewens in 1910 at the Wellcome labs in London, England.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Millar T (2002). Biochemistry explained : a practical guide to learning biochemistry. London: Routledge. p. 40. ISBN 9780415299411. Archived from the original on 15 August 2016.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search