
Back اکتوئین AZB (S)-2-Methyl-3,4,5,6-tetrahydropyrimidin-4-carbonsäure German Ectoína Spanish اکتوئین Persian Ectoïne French Ectoina Italian エクトイン Japanese 엑토인 Korean Ectoïne Dutch Ektoina Polish
| |
| Names | |
|---|---|
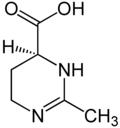
| Preferred IUPAC name
(4S)-2-Methyl-3,4,5,6-tetrahydropyrimidine-4-carboxylic acid | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H10N2O2 | |
| Molar mass | 142.158 g·mol−1 |
| Appearance | White powder |
| Density | 1.568 g/cm3 |
| Soluble in water | |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ectoine (1,4,5,6-tetrahydro-2-methyl-4-pyrimidinecarboxylic acid) is a natural compound found in several species of bacteria. It is a compatible solute which serves as a protective substance by acting as an osmolyte and thus helps organisms survive extreme osmotic stress. Ectoine is found in high concentrations in halophilic microorganisms and confers resistance towards salt and temperature stress. Ectoine was first identified in the microorganism Ectothiorhodospira halochloris,[3][4] but has since been found in a wide range of Gram-negative and Gram-positive bacteria. Other species of bacteria in which ectoine was found include:
- Brevibacterium linens[4]
- Halomonas elongata[3][5]
- Marinococcus halophilus[6]
- Pseudomonas stutzeri[5]
- Halomonas titanicae[7][8]
- Halorhodospira halophila[9]
- Halomonas ventosae[10]
- ^ Ectoine at Sigma-Aldrich
- ^ Ectoine SDS
- ^ a b Peters, P; Miwatani, T; Honda, T (1990). "The biosynthesis of ectoine". FEMS Microbiology Letters. 71 (2): 157–61. doi:10.1016/0378-1097(90)90049-V. PMID 1601286.
- ^ a b Bernard, T.; Jebbar, M.; Rassouli, Y.; Himdi-Kabbab, S.; Hamelin, J.; Blanco, C. (1993). "Ectoine accumulation and osmotic regulation in Brevibacterium linens" (PDF). Journal of General Microbiology. 139: 129–136. doi:10.1099/00221287-139-1-129.
- ^ a b Stöveken, N; Pittelkow, M; Sinner, T; Jensen, R. A.; Heider, J; Bremer, E (2011). "A specialized aspartokinase enhances the biosynthesis of the osmoprotectants ectoine and hydroxyectoine in Pseudomonas stutzeri A1501". Journal of Bacteriology. 193 (17): 4456–68. doi:10.1128/JB.00345-11. PMC 3165526. PMID 21725014.
- ^ Louis, P; Galinski, E. A. (1997). "Characterization of genes for the biosynthesis of the compatible solute ectoine from Marinococcus halophilus and osmoregulated expression in Escherichia coli". Microbiology. 143 ( Pt 4) (4): 1141–9. doi:10.1099/00221287-143-4-1141. PMID 9141677.
- ^ Augenstein, Seth (6 September 2016). "'Extremophile Bacteria' Will Eat Away Wreck of the Titanic". laboratoryequipment.com.
- ^ Zaccai, Giuseppe; Bagyan, Irina; Combet, Jérôme; Cuello, Gabriel J.; Demé, Bruno; Fichou, Yann; Gallat, François-Xavier; Galvan Josa, Victor M.; von Gronau, Susanne; Haertlein, Michael; Martel, Anne; Moulin, Martine; Neumann, Markus; Weik, Martin; Oesterhelt, Dieter (16 August 2016). "Neutrons describe ectoine effects on water H-bonding and hydration around a soluble protein and a cell membrane". Scientific Reports. 6: 31434. Bibcode:2016NatSR...631434Z. doi:10.1038/srep31434. PMC 4985633. PMID 27527336.
- ^ "HAMAP: Halorhodospira halophila (strain DSM 244 / SL1) (Ectothiorhodospira halophila (strain DSM 244 / SL1)) complete proteome ExPASy Proteomics Server. Swiss Institute of Bioinformatics[permanent dead link] http://hamap.expasy.org/proteomes/HALHL.html
- ^ Zhu, Daochen; Niu, Lili; Wang, Chenxiang; Nagata, Shinichi (September 2007). "Isolation and characterisation of moderately halophilic bacteriumHalomonas ventosae DL7 synthesizing ectoine as compatible solute" (PDF). Annals of Microbiology. 57 (3): 401–406. doi:10.1007/BF03175080. S2CID 43421438. Archived from the original (PDF) on 2 October 2020. Retrieved 5 December 2019.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search
