
Back إرغين Arabic ارجین AZB Ergine Danish Ergin German Ergino Esperanto Amida de ácido D-lisérgico Spanish LSA Estonian LSA Finnish Ergine French Ergina Italian
This article needs additional citations for verification. (July 2007) |
 | |
 | |
| Clinical data | |
|---|---|
| Other names | LSA; d-Lysergic acid amide; d-Lysergamide; Ergine; LA-111 |
| Pregnancy category |
|
| Routes of administration | Oral, intramuscular injection |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.841 |
| Chemical and physical data | |
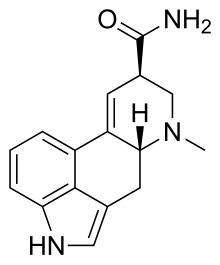
| Formula | C16H17N3O |
| Molar mass | 267.332 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 135 °C (275 °F) Decomposes[4] |
| |
| |
| (verify) | |
Ergine, also known as d-lysergic acid amide (LSA) and d-lysergamide, is an ergoline alkaloid that occurs in various species of vines of the Convolvulaceae and some species of fungi. The psychedelic properties in the seeds of ololiuhqui, Hawaiian baby woodrose and morning glories have been linked to ergine and/or isoergine, its epimer, as it is an alkaloid present in the seeds.[5][6][7]
- ^ Erowid Morning Glory Basics, Erowid.org, retrieved 2012-02-03
- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ^ "Arrêté du 20 mai 2021 modifiant l'arrêté du 22 février 1990 fixant la liste des substances classées comme stupéfiants". www.legifrance.gouv.fr (in French). 20 May 2021.
- ^ Smith S, Timmis GM (1932). "98. The alkaloids of ergot. Part III. Ergine, a new base obtained by the degradation of ergotoxine and ergotinine". Journal of the Chemical Society (Resumed): 763. doi:10.1039/jr9320000763.
- ^ Perrine DM (2000). "Mixing the Kykeon" (PDF). ELEUSIS: Journal of Psychoactive Plants and Compounds. New Series 4: 9. Archived from the original (PDF) on 2019-07-20. Retrieved 2008-05-05.
- ^ Alexander Shulgin, "#26. LSD-25", TiHKAL, Erowid.org, retrieved 2012-02-03
- ^ Hofmann A (2009). LSD My Problem Child: Reflections on Sacred Drugs, Mysticism, and Science (4th ed.). MAPS.org. ISBN 978-0979862229.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search