
Back معالجة بالعاثيات Arabic Фагавая тэрапія Byelorussian Фагавая тэрапія BE-X-OLD Bakteriofagterapi Danish Phagentherapie German Fagoterapia Spanish Faagiterapia Finnish Phagothérapie French Fágterápia Hungarian Terapi fag ID
This article needs additional citations for verification. (February 2023) |

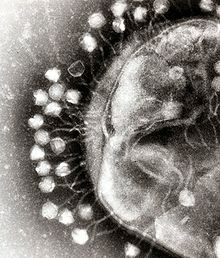
Phage therapy, viral phage therapy, or phagotherapy is the therapeutic use of bacteriophages for the treatment of pathogenic bacterial infections.[1][2][3] This therapeutic approach emerged at the beginning of the 20th century but was progressively replaced by the use of antibiotics in most parts of the world after the Second World War. Bacteriophages, known as phages, are a form of virus[4] that attach to bacterial cells and inject their genome into the cell.[5] The bacteria's production of the viral genome interferes with its ability to function, halting the bacterial infection.[5] The bacterial cell causing the infection is unable to reproduce and instead produces additional phages.[4] Phages are very selective in the strains of bacteria they are effective against.[5]
Advantages include reduced side effects and reduced risk of the bacterium developing resistance, since[5] bacteriophages are much more specific than antibiotics. They are typically harmless not only to the host organism but also to other beneficial bacteria, such as the gut microbiota, reducing the chances of opportunistic infections.[6] They have a high therapeutic index; that is, phage therapy would be expected to give rise to few side effects, even at higher-than-therapeutic levels.[7] Because phages replicate in vivo (in cells of living organism), a smaller effective dose can be used.[8]
Disadvantages include the difficulty of finding an effective phage for a particular infection; a phage will kill a bacterium only if it matches the specific strain.[5] However, virulent phages can be isolated much more easily than other compounds and natural products.[8] Consequently, phage mixtures ("cocktails") are sometimes used to improve the chances of success.[9] Alternatively, samples taken from recovering patients sometimes contain appropriate phages that can be grown to cure other patients infected with the same strain.[10] Ongoing challenges include the need to increase phage collections from reference phage banks, the development of efficient phage screening methods for the fast identification of the therapeutic phage(s), the establishment of efficient phage therapy strategies to tackle infectious biofilms, the validation of feasible phage production protocols that assure quality and safety of phage preparations, and the guarantee of stability of phage preparations during manufacturing, storage, and transport.
Phages tend to be more successful than antibiotics where there is a biofilm covered by a polysaccharide layer, which antibiotics typically cannot penetrate.[11] Phage therapy can disperse the biofilm generated by antibiotic-resistant bacteria.[12] However, the interactions between phages and biofilms can be complex, with phages developing symbiotic as well as predatory relationships with biofilms.[9]
Phages are currently being used therapeutically to treat bacterial infections that do not respond to conventional antibiotics,[2][1][13] particularly in Russia[14] and Georgia.[15][16][17] There is also a phage therapy unit in Wrocław, Poland, established in 2005, which continues several-decades-long research by the Institute of Immunology and Experimental Therapy of the Polish Academy of Sciences, the only such centre in a European Union country.[18] Phages are the subject of renewed clinical attention in Western countries, such as the United States. In 2019, the United States Food and Drug Administration approved the first US clinical trial for intravenous phage therapy.[19]
Phage therapy has many potential applications in human medicine as well as dentistry, veterinary science, and agriculture.[20] If the target host of a phage therapy treatment is not an animal, the term "biocontrol" (as in phage-mediated biocontrol of bacteria) is usually employed, rather than "phage therapy".[9]
- ^ a b Kortright KE, Chan BK, Koff JL, Turner PE (February 2019). "Phage Therapy: A Renewed Approach to Combat Antibiotic-Resistant Bacteria". Cell Host & Microbe. 25 (2): 219–232. doi:10.1016/j.chom.2019.01.014. PMID 30763536. S2CID 73439131.
- ^ a b Gordillo Altamirano FL, Barr JJ (April 2019). "Phage Therapy in the Postantibiotic Era". Clinical Microbiology Reviews. 32 (2). doi:10.1128/CMR.00066-18. PMC 6431132. PMID 30651225.
- ^ Cite error: The named reference
Kohnwas invoked but never defined (see the help page). - ^ a b Sulakvelidze A, Alavidze Z, Morris JG (March 2001). "Bacteriophage therapy". Antimicrobial Agents and Chemotherapy. 45 (3): 649–659. doi:10.1128/AAC.45.3.649-659.2001. PMC 90351. PMID 11181338.
- ^ a b c d e Srisuknimit V (1 February 2018). "Bacteriophage: A solution to our antibiotics problem? How we can us a virus to fight bacterial infection". Science in the News. Retrieved 28 January 2020.
- ^ Cite error: The named reference
phagewas invoked but never defined (see the help page). - ^ Fernández L, Gutiérrez D, García P, Rodríguez A (August 2019). "The Perfect Bacteriophage for Therapeutic Applications – A Quick Guide". Antibiotics. 8 (3): 126. doi:10.3390/antibiotics8030126. PMC 6783975. PMID 31443585.
- ^ a b Saha D, Mukherjee R (July 2019). "Ameliorating the antimicrobial resistance crisis: phage therapy". IUBMB Life. 71 (7): 781–790. doi:10.1002/iub.2010. PMID 30674079.
- ^ a b c Pires DP, Melo LD, Azeredo J (September 2021). "Understanding the Complex Phage-Host Interactions in Biofilm Communities". Annual Review of Virology. 8 (1): 73–94. doi:10.1146/annurev-virology-091919-074222. hdl:1822/74528. PMID 34186004.
- ^ Hyman P (March 2019). "Phages for Phage Therapy: Isolation, Characterization, and Host Range Breadth". Pharmaceuticals. 12 (1): 35. doi:10.3390/ph12010035. PMC 6469166. PMID 30862020.
- ^ Cite error: The named reference
LabNewswas invoked but never defined (see the help page). - ^ Pai L, Patil S, Liu S, Wen F (2023). "A growing battlefield in the war against biofilm-induced antimicrobial resistance: insights from reviews on antibiotic resistance". Front Cell Infect Microbiol. 13: 1327069. doi:10.3389/fcimb.2023.1327069. PMC 10770264. PMID 38188636.
- ^ González-Mora A, Hernández-Pérez J, Iqbal HM, Rito-Palomares M, Benavides J (September 2020). "Bacteriophage-Based Vaccines: A Potent Approach for Antigen Delivery". Vaccines. 8 (3): 504. doi:10.3390/vaccines8030504. PMC 7565293. PMID 32899720.
- ^ Cite error: The named reference
Eaters of bacteria: Is phage therapy ready for the big time?was invoked but never defined (see the help page). - ^ Cite error: The named reference
horizonwas invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid15986542was invoked but never defined (see the help page). - ^ Cite error: The named reference
Thielwas invoked but never defined (see the help page). - ^ "Phage Therapy Unit of the Medical Centre of the Institute of Immunology and Experimental Therapy PAS". Instytut Immunologii i Terapii Doświadczalnej im. Ludwika Hirszfelda Polskiej Akademii Nauk.
- ^ Jones H (11 January 2019). "First US clinical trial for intravenous phage therapy gets FDA approval". Biotechniques. Archived from the original on 28 September 2020. Retrieved 28 January 2020.
- ^ Cite error: The named reference
McAuliffewas invoked but never defined (see the help page).
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search