
Back حمض الرتينويك Arabic رتینوئیک اسید AZB Retinoidna kiselina BS Àcid retinoic Catalan Retinsäuren German Ácido retinoico Spanish Azido erretinoiko Basque رتینوئیک اسید Persian Acide rétinoïque French Ácido retinoico Galician
| |

| |
| Names | |
|---|---|
| IUPAC name
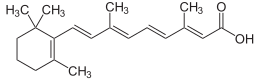
Retinoic acid
| |
| Systematic IUPAC name
(2E,4E,6E,8E)-3,7-Dimethyl-9-(2,6,6-trimethylcyclohex-1-en-1-yl)nona-2,4,6,8-tetraenoic acid | |
| Other names
vitamin A acid; RA
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C20H28O2 | |
| Molar mass | 300.43512 g/mol |
| Appearance | yellow to light orange crystalline powder with a characteristic of a floral scent[1] |
| Melting point | 180 to 182 °C (356 to 360 °F; 453 to 455 K) crystals from ethanol[1] |
| nearly insoluble | |
| Solubility in fat | soluble |
| Related compounds | |
Related compounds
|
retinol; retinal; beta-carotene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Retinoic acid (used simplified here for all-trans-retinoic acid) is a metabolite of vitamin A1 (all-trans-retinol) that mediates the functions of vitamin A1 required for growth and development. All-trans-retinoic acid is required in chordate animals, which includes all higher animals from fish to humans. During early embryonic development, all-trans-retinoic acid generated in a specific region of the embryo helps determine position along the embryonic anterior/posterior axis by serving as an intercellular signaling molecule that guides development of the posterior portion of the embryo.[2] It acts through Hox genes, which ultimately control anterior/posterior patterning in early developmental stages.[3]
All-trans-retinoic acid (ATRA) is the major occurring retinoic acid, while isomers like 13-cis- and 9-cis-retinoic acid are also present in much lower levels.[4]
The key role of all-trans-retinoic acid in embryonic development mediates the high teratogenicity of retinoid pharmaceuticals, such as isotretinoin (13-cis-retinoic acid) used for treatment of cancer and acne. Oral megadoses of preformed vitamin A (retinyl palmitate), and all-trans-retinoic acid itself, also have teratogenic potential by this same mechanism.
- ^ a b Merck Index, 13th Edition, 8251.
- ^ Duester G (September 2008). "Retinoic acid synthesis and signaling during early organogenesis". Cell. 134 (6): 921–31. doi:10.1016/j.cell.2008.09.002. PMC 2632951. PMID 18805086.
- ^ Holland LZ (May 2007). "Developmental biology: a chordate with a difference". Nature. 447 (7141): 153–5. Bibcode:2007Natur.447..153H. doi:10.1038/447153a. PMID 17495912. S2CID 5549210.
- ^ Rühl R, Krezel W, de Lera AR (December 2018). "9-Cis-13,14-dihydroretinoic acid, a new endogenous mammalian ligand of retinoid X receptor and the active ligand of a potential new vitamin A category: vitamin A5". Nutrition Reviews. 76 (12): 929–941. doi:10.1093/nutrit/nuy057. PMID 30358857.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search