
Back فلوريد الفاناديوم الخماسي Arabic فلوئورید وانادیوم (V) AZB Fluorid vanadičný Czech Vanadium(V)-fluorid German فلوئورید وانادیم (V) Persian Vanadiinipentafluoridi Finnish Vanádium-pentafluorid Hungarian Pentafluoruro di vanadio Italian フッ化バナジウム(V) Japanese Fluoreto de vanádio(V) Portuguese
| |
| Names | |
|---|---|
| IUPAC name
Vanadium(V) fluoride
| |
| Other names
Vanadium pentafluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.029.112 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
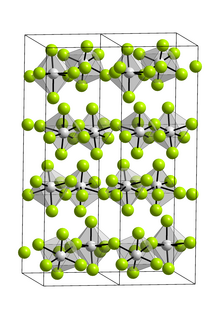
| VF5 | |
| Molar mass | 145.934 |
| Appearance | colorless solid |
| Density | 2.502 g/cm3 (solid) |
| Melting point | 19.5 °C (67.1 °F; 292.6 K) |
| Boiling point | 48.3 °C (118.9 °F; 321.4 K) |
| Related compounds | |
Other cations
|
Niobium(V) fluoride Tantalum(V) fluoride |
Related Vanadium compounds
|
Vanadium(V) oxide Vanadium trifluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Vanadium(V) fluoride is the inorganic compound with the chemical formula VF5. It is a colorless volatile liquid[1] that freezes near room temperature. It is a highly reactive compound, as indicated by its ability to fluorinate organic substances.[2]
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 989. ISBN 978-0-08-037941-8.
- ^ Canterford, J. H.; O'Donnell, Thomas A. (1967-03-01). "Reactivity of transition metal fluorides. IV. Oxidation-reduction reactions of vanadium pentafluoride". Inorganic Chemistry. 6 (3): 541–544. doi:10.1021/ic50049a025. ISSN 0020-1669.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search