Back زاليبلون Arabic Saleplon Welsh Zaleplon German Zaleplon Spanish زالپلون Persian Tsaleploni Finnish Zaleplon French Zaleplon Hungarian ଜାଲେପ୍ଲୋନ OR Zaleplon Polish
This article needs additional citations for verification. (June 2018) |
| |||
| Clinical data | |||
|---|---|---|---|
| Trade names | Sonata, others | ||
| AHFS/Drugs.com | Monograph | ||
| MedlinePlus | a601251 | ||
| License data | |||
| Addiction liability | Moderate | ||
| Routes of administration | By mouth | ||
| Drug class | nonbenzodiazepine | ||
| ATC code | |||
| Legal status | |||
| Legal status | |||
| Pharmacokinetic data | |||
| Bioavailability | 30% (oral)[4] | ||
| Metabolism | Liver aldehyde oxidase (91%), CYP3A4 (9%)[5] | ||
| Elimination half-life | 1 hr[4] | ||
| Excretion | Kidney | ||
| Identifiers | |||
| |||
| CAS Number | |||
| PubChem CID | |||
| IUPHAR/BPS | |||
| DrugBank | |||
| ChemSpider | |||
| UNII | |||
| KEGG | |||
| ChEBI | |||
| ChEMBL | |||
| CompTox Dashboard (EPA) | |||
| ECHA InfoCard | 100.126.674 | ||
| Chemical and physical data | |||
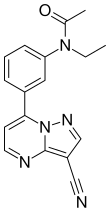
| Formula | C17H15N5O | ||
| Molar mass | 305.341 g·mol−1 | ||
| 3D model (JSmol) | |||
| |||
| |||
| (verify) | |||
Zaleplon, sold under the brand name Sonata among others, is a sedative and hypnotic which is used to treat insomnia. It is a nonbenzodiazepine or Z-drug of the pyrazolopyrimidine class.[6] It was developed by King Pharmaceuticals and approved for medical use in the United States in 1999.[3]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b "Sonata (zaleplon) Capsules CIV". DailyMed. Retrieved 21 March 2023.
- ^ a b Rosen AS, Fournié P, Darwish M, Danjou P, Troy SM (April 1999). "Zaleplon pharmacokinetics and absolute bioavailability". Biopharmaceutics & Drug Disposition. 20 (3): 171–175. doi:10.1002/(sici)1099-081x(199904)20:3<171::aid-bdd169>3.0.co;2-k. PMID 10211871.
- ^ "20859 S009, 011 FDA Approved Labeling Text 12.10.07" (PDF). FDA. Retrieved 21 March 2023.
- ^ Elie R, Rüther E, Farr I, Emilien G, Salinas E (August 1999). "Sleep latency is shortened during 4 weeks of treatment with zaleplon, a novel nonbenzodiazepine hypnotic. Zaleplon Clinical Study Group". The Journal of Clinical Psychiatry. 60 (8): 536–44. doi:10.4088/JCP.v60n0806. PMID 10485636.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search