
Back أسينابين Arabic Asenapin German Ασεναπίνη Greek آسناپین Persian Asenapiini Finnish Asénapine French ଆସେନାପାଇନ OR Asenapina Polish Asenapina Portuguese Asenapină Romanian
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Saphris, Sycrest, Secuado |
| Other names | ORG-5222 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a610015 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Sublingual, transdermal |
| Drug class | Atypical antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 35% (sublingual), <2% (oral),[5][6][3][7] transdermal bioavailability is significantly higher than sublingual[8][9] |
| Protein binding | 95%[5][6][3][7] |
| Metabolism | hepatic (glucurinodation by UGT1A4 and oxidative metabolism by CYP1A2)[5][6][3][7] |
| Elimination half-life | 24 hours (sublingual),[5][6][3][7] 30 hours (transdermal),[8] 33.9 hours (transdermal)[9] |
| Excretion | Kidney (50%), Faecal (40%; ~5–16% as unchanged drug in faeces)[5][6][3][7] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI |
|
| ChEMBL |
|
| ECHA InfoCard | 100.059.828 |
| Chemical and physical data | |
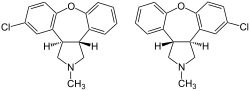
| Formula | C17H16ClNO |
| Molar mass | 285.77 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Asenapine, sold under the brand name Saphris among others, is an atypical antipsychotic medication used to treat schizophrenia and acute mania associated with bipolar disorder as well as the medium to long-term management of bipolar disorder.[4][10]
It was chemically derived via altering the chemical structure of the tetracyclic (atypical) antidepressant, mianserin.[11]
It was initially approved in the United States in 2009[12] and approved as a generic medication in 2020.[13]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d e f "Sycrest 5mg sublingual tablets - Summary of Product Characteristics (SmPC)". (emc). Retrieved 9 September 2020.
- ^ a b "Sycrest EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 9 September 2020.
- ^ a b c d e "Product Information Saphris (asenapine maleate)" (PDF). TGA eBusiness Services. Merck Sharp & Dohme (Australia) Pty Limited. 14 January 2013. Retrieved 23 October 2013.
- ^ a b c d e "Saphris (asenapine maleate) tablet". DailyMed. Organon Pharmaceuticals. March 2013. Retrieved 23 October 2013.
- ^ a b c d e "Product information Sycrest – EMEA/H/C/001177 –II/0012" (PDF). European Medicines Agency. N.V. Organon. 21 February 2013. Archived from the original (PDF) on 28 July 2017. Retrieved 23 October 2013.
- ^ a b Cite error: The named reference
Carrithers 2020was invoked but never defined (see the help page). - ^ a b Suzuki K, Castelli M, Komaroff M, Starling B, Terahara T, Citrome L (2021-03-16). "Pharmacokinetic Profile of the Asenapine Transdermal System (HP-3070)". Journal of Clinical Psychopharmacology. 41 (3): 286–294. doi:10.1097/JCP.0000000000001383. PMC 8083160. PMID 33734167.
- ^ Cite error: The named reference
pmid23719049was invoked but never defined (see the help page). - ^ Minassian A, Young JW (August 2010). "Evaluation of the clinical efficacy of asenapine in schizophrenia". Expert Opinion on Pharmacotherapy. 11 (12): 2107–2115. doi:10.1517/14656566.2010.506188. PMC 2924192. PMID 20642375.
- ^ "Saphris (asenapine) prescribing information" (PDF). Schering Corporation. 2009-08-01. Archived from the original (PDF) on 2009-11-22. Retrieved 2009-09-05.
- ^ "First Generic Drug Approvals". U.S. Food and Drug Administration (FDA). Retrieved 13 February 2021.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search