
Back بيكولينات الكروم الثلاثي Arabic پیکولینات کوروم (III) AZB Chrom(III)-picolinat German پیکولینات کروم (III) Persian Kromi(III)pikolinaatti Finnish Cromo picolinato Italian Picolinato de cromo(III) Portuguese Hrom(III) pikolinat Serbo-Croatian Hrom(III) pikolinat Serbian Krompikolinat Swedish
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
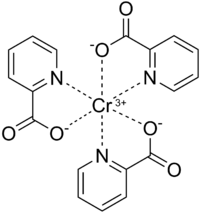
Tris(picolinate)chromium(III)
| |||
| Identifiers | |||
3D model (JSmol)
|
| ||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.131.423 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| Cr(C6H4NO2)3 | |||
| Molar mass | 418.33 g/mol | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Chromium(III) picolinate is a chemical compound with the formula Cr(C5H4N(CO2H))3, commonly abbreviated as CrPic3. It is sold as a nutritional supplement to treat type 2 diabetes and promote weight loss.[1] This bright-red coordination compound is derived from chromium(III) and picolinic acid. Trace amounts (25-35 mcg[2]) of chromium are needed for glucose utilization by insulin in normal health, but deficiency is extremely uncommon and has been observed usually in people receiving 100% of their nutrient needs intravenously, i.e., total parenteral nutrition diets.[3] Chromium has been identified as regulating insulin by increasing the sensitivity of the insulin receptor.[4] As such, chromium(III) picolinate has been proposed as a treatment for type 2 diabetes, although its effectiveness remains controversial due to conflicting evidence from human trials.[5]
- ^ Preuss, H. G.; Echard, B.; Perricone, N. V.; Bagchi, D.; Yasmin, T.; Stohs, S. J. (2008). "Comparing metabolic effects of six different commercial trivalent chromium compounds". Journal of Inorganic Biochemistry. 102 (11): 1986–1990. doi:10.1016/j.jinorgbio.2008.07.012. PMID 18774175.
- ^ "The Nutrition Source: Chromium". School of Public Health, Harvard University.
- ^ Review of Chromium Archived February 7, 2012, at the Wayback Machine Expert Group on Vitamins and Minerals Review of Chromium, 12 August 2002
- ^ Stearns DM (2000). "Is chromium a trace essential metal?". BioFactors. 11 (3): 149–62. doi:10.1002/biof.5520110301. PMID 10875302. S2CID 19417496.
- ^ Vincent, John (2010). "Chromium: celebrating 50 years as an essential element?". Dalton Transactions. 39 (16): 3787–3794. doi:10.1039/B920480F. PMID 20372701.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search

