
Back ألدهيد القرفة Arabic سینامالدهید AZB Cinamaldehid Catalan Cinnamaldehyd Czech Zimtaldehyd German Cinamaldehido Esperanto Cinamaldehído Spanish Kaneelaldehüüd Estonian سینامالدهید Persian Kanelialdehydi Finnish
| |
| |

| |
| Names | |
|---|---|
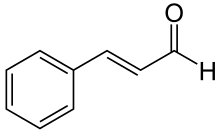
| Preferred IUPAC name
(2E)-3-Phenylprop-2-enal | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| 3DMet | |
| 1071571 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.111.079 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H8O | |
| Molar mass | 132.16 g/mol |
| Appearance | Yellow oil |
| Odor | Pungent, cinnamon-like |
| Density | 1.0497 g/mL |
| Melting point | −7.5 °C (18.5 °F; 265.6 K) |
| Boiling point | 248 °C (478 °F; 521 K) |
| Slightly soluble | |
| Solubility |
|
| −7.48×10−5 cm3/mol | |
Refractive index (nD)
|
1.6195 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H317, H319, H335 | |
| P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P333+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 71 °C (160 °F; 344 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
3400 mg/kg (rat, oral) |
| Related compounds | |
Related compounds
|
Cinnamic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cinnamaldehyde is an organic compound with the formula or C₆H₅CH=CHCHO. Occurring naturally as predominantly the trans (E) isomer, it gives cinnamon its flavor and odor.[1] It is a phenylpropanoid that is naturally synthesized by the shikimate pathway.[2] This pale yellow, viscous liquid occurs in the bark of cinnamon trees and other species of the genus Cinnamomum. The essential oil of cinnamon bark is about 90% cinnamaldehyde.[3] Cinnamaldehyde decomposes to styrene because of oxidation as a result of bad storage or transport conditions. Styrene especially forms in high humidity and high temperatures. This is the reason why cinnamon contains small amounts of styrene.[4]
- ^ "Cinnamon". Transport Information Service. Gesamtverband der Deutschen Versicherungswirtschaft e.V. Retrieved 2007-10-23.
- ^ Gutzeit, Herwig (2014). Plant Natural Products: Synthesis, Biological Functions and Practical Applications. Wiley. pp. 19–21. ISBN 978-3-527-33230-4.
- ^ PubChem. "Cinnamaldehyde". pubchem.ncbi.nlm.nih.gov. Retrieved 2019-10-18.
- ^ "High daily intakes of cinnamon: Health risk cannot be ruled out" (PDF). Federal Institute for Risk Assessment (BfR). 18 August 2006. Archived (PDF) from the original on 7 March 2022. Retrieved 20 May 2022.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search

