
Back زراعة العظام Arabic İmplantlar Azerbaijani Медицински имплант Bulgarian Implant (medicina) Catalan Implantat German Enplantaĵo Esperanto Implante (medicina) Spanish Implantaat Estonian ایمپلنت Persian Implantti Finnish

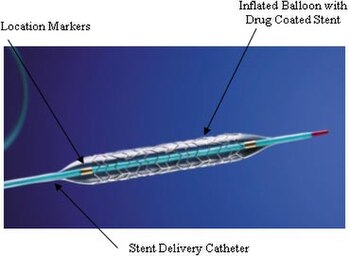
An implant is a medical device manufactured to replace a missing biological structure, support a damaged biological structure, or enhance an existing biological structure. For example, an implant may be a rod, used to strengthen weak bones.[1][2] Medical implants are human-made devices, in contrast to a transplant, which is a transplanted biomedical tissue. The surface of implants that contact the body might be made of a biomedical material such as titanium, silicone, or apatite depending on what is the most functional.[3] In 2018, for example, American Elements developed a nickel alloy powder for 3D printing robust, long-lasting, and biocompatible medical implants.[4] In some cases implants contain electronics, e.g. artificial pacemaker and cochlear implants. Some implants are bioactive, such as subcutaneous drug delivery devices in the form of implantable pills or drug-eluting stents.[5]
- ^ Kong, L.; Heydari, Z.; Lami, G.H.; Saberi, A.; Baltatu, M.S.; Vizureanu, P. A Comprehensive Review of the Current Research Status of Biodegradable Zinc Alloys and Composites for Biomedical Applications. Materials 2023, 16, 4797. https://doi.org/10.3390/ma16134797 PMID: 37445111
- ^ Zhao, J.; Haowei, M.; Saberi, A.; Heydari, Z.; Baltatu, M.S. Carbon Nanotube (CNT) Encapsulated Magnesium-Based Nanocomposites to Improve Mechanical, Degradation and Antibacterial Performances for Biomedical Device Applications. Coatings 2022, 12, 1589. https://doi.org/10.3390/coatings12101589
- ^ Wong, J.Y.; Bronzino, J.D.; Peterson, D.R., eds. (2012). Biomaterials: Principles and Practices. Boca Raton, Florida: CRC Press. p. 281. ISBN 9781439872512. Retrieved 12 March 2016.
- ^ "Medical Devices". American Elements. Retrieved 20 December 2023.
- ^ "Download Product Code Classification Files". FDA.org/medicaldevices. Food and Drug Administration. 4 November 2014. Retrieved 12 March 2016.
Relevant info in the foiclass.zip file.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search