
Back Molekulêre orbitaal Afrikaans Orbital molecular AN مدار جزيئي Arabic Orbital molecular AST আণবিক কক্ষক Bengali/Bangla Molekulska orbitala BS Orbital molecular Catalan Molekulový orbital Czech Μοριακό τροχιακό Greek Orbital molecular Spanish
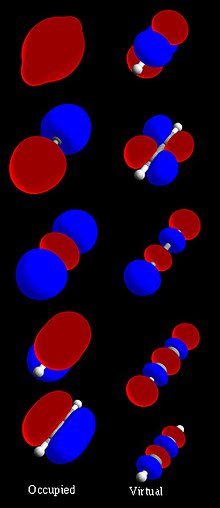
In chemistry, a molecular orbital (/ɒrbədl/) is a mathematical function describing the location and wave-like behavior of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of finding an electron in any specific region. The terms atomic orbital and molecular orbital[a] were introduced by Robert S. Mulliken in 1932 to mean one-electron orbital wave functions.[2] At an elementary level, they are used to describe the region of space in which a function has a significant amplitude.
In an isolated atom, the orbital electrons' location is determined by functions called atomic orbitals. When multiple atoms combine chemically into a molecule by forming a valence chemical bond, the electrons' locations are determined by the molecule as a whole, so the atomic orbitals combine to form molecular orbitals. The electrons from the constituent atoms occupy the molecular orbitals. Mathematically, molecular orbitals are an approximate solution to the Schrödinger equation for the electrons in the field of the molecule's atomic nuclei. They are usually constructed by combining atomic orbitals or hybrid orbitals from each atom of the molecule, or other molecular orbitals from groups of atoms. They can be quantitatively calculated using the Hartree–Fock or self-consistent field (SCF) methods.
Molecular orbitals are of three types: bonding orbitals which have an energy lower than the energy of the atomic orbitals which formed them, and thus promote the chemical bonds which hold the molecule together; antibonding orbitals which have an energy higher than the energy of their constituent atomic orbitals, and so oppose the bonding of the molecule, and non-bonding orbitals which have the same energy as their constituent atomic orbitals and thus have no effect on the bonding of the molecule.
- ^ orbital. Merriam-Webster. Retrieved April 18, 2021.
{{cite book}}:|work=ignored (help) - ^ a b Mulliken, Robert S. (July 1932). "Electronic Structures of Polyatomic Molecules and Valence. II. General Considerations". Physical Review. 41 (1): 49–71. Bibcode:1932PhRv...41...49M. doi:10.1103/PhysRev.41.49.
- ^ Brown, Theodore (2002). Chemistry : the central science. Upper Saddle River, NJ: Prentice Hall. ISBN 0-13-066997-0.
Cite error: There are <ref group=lower-alpha> tags or {{efn}} templates on this page, but the references will not show without a {{reflist|group=lower-alpha}} template or {{notelist}} template (see the help page).
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search