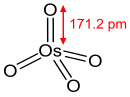Back Osmium(VIII)oksied Afrikaans أكسيد الأوزميوم الثماني Arabic اوسمیوم تتراوکسید AZB Tetraòxid d'osmi Catalan Oxid osmičelý Czech Osmium(VIII)-oxid German Tetróxido de osmio Spanish اسمیم تتراکسید Persian Osmiumtetroksidi Finnish Tétroxyde d'osmium French
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Osmium tetraoxide | |||
| Systematic IUPAC name
Tetraoxoosmium | |||
| Other names
Osmium(VIII) oxide
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.040.038 | ||
| EC Number |
| ||
| MeSH | Osmium+tetroxide | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | UN 2471 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| OsO4 | |||
| Molar mass | 254.23 g/mol | ||
| Appearance | White volatile solid | ||
| Odor | Acrid, chlorine-like | ||
| Density | 4.9 g/cm3[1] | ||
| Melting point | 40.25 °C (104.45 °F; 313.40 K) | ||
| Boiling point | 129.7[2] °C (265.5 °F; 402.8 K) | ||
| 5.70 g/100 mL (10 °C) 6.23 g/100 mL (25 °C) | |||
| Solubility | Soluble in most organic solvents, ammonium hydroxide, phosphorus oxychloride | ||
| Solubility in CCl4 | 375 g/100 mL | ||
| Vapor pressure | 7 mmHg (20 °C)[3] | ||
| Structure[4] | |||
| Monoclinic, mS20 | |||
| C2/c | |||
α = 90°, β = 116.58°, γ = 90°
| |||
Lattice volume (V)
|
326.8 Å3 | ||
Formula units (Z)
|
4 | ||
| tetrahedral | |||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H300, H310, H314, H330 | |||
| P260, P262, P264, P270, P271, P280, P284, P301+P310, P301+P330+P331, P302+P350, P303+P361+P353, P304+P340, P305+P351+P338, P310, P320, P321, P322, P330, P361, P363, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
1316 mg/m3 (rabbit, 30 min) 423 mg/m3 (rat, 4 hr) 423 mg/m3 (mouse, 4 hr)[5] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 0.002 mg/m3[3] | ||
REL (Recommended)
|
TWA 0.002 mg/m3 (0.0002 ppm) ST 0.006 mg/m3 (0.0006 ppm)[3] | ||
IDLH (Immediate danger)
|
1 mg/m3[3] | ||
| Safety data sheet (SDS) | ICSC 0528 | ||
| Related compounds | |||
Other cations
|
Ruthenium tetroxide Hassium tetroxide | ||
| Osmium(IV) oxide | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Osmium tetroxide (also osmium(VIII) oxide) is the chemical compound with the formula OsO4. The compound is noteworthy for its many uses, despite its toxicity and the rarity of osmium. It also has a number of unusual properties, one being that the solid is volatile. The compound is colourless, but most samples appear yellow.[6] This is most likely due to the presence of the impurity OsO2, which is yellow-brown in colour.[7] In biology, its property of binding to lipids has made it a widely-used stain in electron microscopy.
- ^ "Osmium tetroxide ICSC: 0528". InChem.
- ^ Koda, Yoshio (1986). "Boiling Points and Ideal Solutions of Ruthenium and Osmium Tetraoxides". Journal of the Chemical Society, Chemical Communications. 1986 (17): 1347–1348. doi:10.1039/C39860001347.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0473". National Institute for Occupational Safety and Health (NIOSH).
- ^ Krebs, B.; Hasse, K. D. (1976). "Refinements of the Crystal Structures of KTcO4, KReO4 and OsO4. The Bond Lengths in Tetrahedral Oxo-Anions and Oxides of d0 Transition Metals". Acta Crystallographica B. 32 (5): 1334–1337. doi:10.1107/S056774087600530X.
- ^ "Osmium tetroxide (as Os)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Girolami, Gregory (2012). "Osmium weighs". Nature Chemistry. 4 (11): 954. Bibcode:2012NatCh...4..954G. doi:10.1038/nchem.1479. PMID 23089872.
- ^ Cotton and Wilkinson, Advanced Inorganic Chemistry, p.1002
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search