Back سداسي فلوريد السيلينيوم Arabic هقزافلوئورید سلنیوم AZB Fluorid selenový Czech Selenhexafluorid German Hexafluoruro de selenio Spanish هگزافلوئورید سلنیم Persian Seleeniheksafluoridi Finnish Hexafluorure de sélénium French Esafluoruro di selenio Italian 六フッ化セレン Japanese
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Selenium hexafluoride
| |||
| Other names
Selenium(VI) fluoride, Selenium fluoride
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.149.506 | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
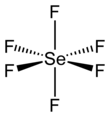
| SeF6 | |||
| Molar mass | 192.9534 g/mol | ||
| Appearance | colourless gas | ||
| Density | 0.007887 g/cm3[1] | ||
| Melting point | −39 °C (−38 °F; 234 K) | ||
| Boiling point | −34.5 °C (−30.1 °F; 238.7 K) sublimes | ||
| insoluble | |||
| Vapor pressure | >1 atm (20°C)[2] | ||
| −51.0·10−6 cm3/mol | |||
Refractive index (nD)
|
1.895 | ||
| Structure | |||
| Orthorhombic, oP28 | |||
| Pnma, No. 62 | |||
| octahedral (Oh) | |||
| 0 | |||
| Thermochemistry | |||
Std enthalpy of
formation (ΔfH⦵298) |
-1030 kJ/mol[3] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
toxic, corrosive | ||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
10 ppm (rat, 1 hr) 10 ppm (mouse, 1 hr) 10 ppm (guinea pig, 1 hr)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 0.05 ppm (0.4 mg/m3)[2] | ||
REL (Recommended)
|
TWA 0.05 ppm[2] | ||
IDLH (Immediate danger)
|
2 ppm[2] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Selenium hexafluoride is the inorganic compound with the formula SeF6. It is a very toxic colourless gas described as having a "repulsive" odor.[5] It is not widely encountered and has no commercial applications.[6]
- ^ Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0551". National Institute for Occupational Safety and Health (NIOSH).
- ^ Wiberg, E.; Holleman, A. F. (2001). Inorganic Chemistry. Elsevier. ISBN 0-12-352651-5.
- ^ "Selenium hexafluoride". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "Material Safety" (PDF). Retrieved 2010-07-24.
- ^ Langner, B. E. "Selenium and Selenium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a23_525. ISBN 978-3527306732.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search