
Back فوسفات الحديد والليثيوم Arabic لیتیوم دمیر فوسفات AZB Fosfat de ferro liti Catalan Lithiumeisenphosphat German لیتیم آهن فسفات Persian Phosphate de fer et de lithium French Litio ferro fosfato Italian Litijum gvožđe fosfat Serbo-Croatian Litijum gvožđe fosfat Serbian இலித்தியம் இரும்பு பாசுபேட்டு Tamil
| |
| |
| Names | |
|---|---|
| IUPAC name
iron(2+) lithium phosphate (1:1:1)
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.124.705 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
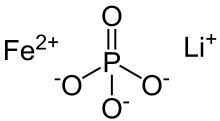
| FeLiO 4P | |
| Molar mass | 157.757 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
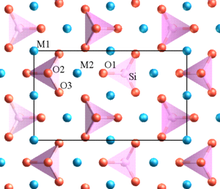
Lithium iron phosphate or lithium ferro-phosphate (LFP) is an inorganic compound with the formula LiFePO
4. It is a gray, red-grey, brown or black solid that is insoluble in water. The material has attracted attention as a component of lithium iron phosphate batteries,[1] a type of Li-ion battery. This battery chemistry is targeted for use in power tools, electric vehicles, solar energy installations[2] and more recently large grid-scale energy storage.[3]
Most lithium batteries (Li-ion) used in consumer electronics products use cathodes made of lithium compounds such as lithium cobalt oxide (LiCoO
2), lithium manganese oxide (LiMn
2O
4), and lithium nickel oxide (LiNiO
2). The anodes are generally made of graphite.
Lithium iron phosphate exists naturally in the form of the mineral triphylite, but this material has insufficient purity for use in batteries.
- ^ Park, O. K.; Cho, Y.; Lee, S.; Yoo, H.-C.; Song, H.-K.; Cho, J., "Who Will Drive Electric Vehicles, Olivine or Spinel?", Energy Environ. Sci. 2011, volume 4, pages 1621-1633. doi:10.1039/c0ee00559b
- ^ Ozawa, Ryan (7 July 2015). "New Energy Storage Startup to Take Hawaii Homes Off-Grid". Hawaii Blog. Retrieved 2015-07-09.
- ^ "Google Looks to Batteries as Replacement for Diesel Generators". 16 December 2020.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search