
Back سيلوسين Arabic سیلوسین AZB Psilocin Czech Psilocin German Psilocino Esperanto Psilocina Spanish Psilozina Basque سیلوسین Persian Psilosiini Finnish Psilocine French
 | |
 | |
| Clinical data | |
|---|---|
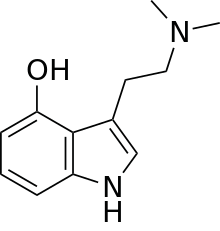
| Other names | 4-hydroxy-N,N-dimethyltryptamine |
| Routes of administration | Oral, IV |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | 1-3 hours[2] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.007.543 |
| Chemical and physical data | |
| Formula | C12H16N2O |
| Molar mass | 204.273 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 173 to 176 °C (343 to 349 °F) |
| |
| |
| | |
Psilocin (also known as 4-HO-DMT, 4-hydroxy DMT, psilocine, psilocyn, or psilotsin) is a substituted tryptamine alkaloid and a serotonergic psychedelic substance. It is present in most psychedelic mushrooms[3] together with its phosphorylated counterpart psilocybin. Psilocin is a Schedule I drug under the Convention on Psychotropic Substances.[4] Acting on the 5-HT2A receptors, psilocin modulates the production and reuptake of serotonin.[5] The mind-altering effects of psilocin are highly variable and subjective and resemble those of LSD and DMT.
- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ^ Tylš F, Páleníček T, Horáček J (March 2014). "Psilocybin--summary of knowledge and new perspectives". European Neuropsychopharmacology. 24 (3): 342–356. doi:10.1016/j.euroneuro.2013.12.006. PMID 24444771. S2CID 10758314.
- ^ Gotvaldová K, Borovička J, Hájková K, Cihlářová P, Rockefeller A, Kuchař M (November 2022). "Extensive Collection of Psychotropic Mushrooms with Determination of Their Tryptamine Alkaloids". International Journal of Molecular Sciences. 23 (22): 14068. doi:10.3390/ijms232214068. PMC 9693126. PMID 36430546.
- ^ "List of psychotropic substances under international control" (PDF) (23rd ed.). Vienna Austria: International Narcotics Control Board. August 2003. Archived from the original (PDF) on 4 February 2012. Retrieved 2012-10-11.
- ^ Madsen MK, Fisher PM, Burmester D, Dyssegaard A, Stenbæk DS, Kristiansen S, et al. (June 2019). "Psychedelic effects of psilocybin correlate with serotonin 2A receptor occupancy and plasma psilocin levels". Neuropsychopharmacology. 44 (7): 1328–1334. doi:10.1038/s41386-019-0324-9. PMC 6785028. PMID 30685771.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search