
Back ایندوفنول AZB Indophenol German Indofenol Spanish ایندوفنول Persian Indofenoli Finnish Indophénol French Indofenolo Italian Indofenol Portuguese Индофенолы Russian Indofenol Serbo-Croatian
| |
| |

| |
| Names | |
|---|---|
| IUPAC name
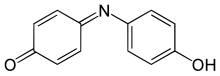
4-(4-hydroxyphenyl)iminocyclohexa-2,5-dien-1-one
| |
| Other names
Benzenoneindophenol, phenolindophenol[1]
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.007.194 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H9NO2 | |
| Molar mass | 199.209 g·mol−1 |
| Appearance | Reddish-blue powder[1] |
| Melting point | above 300 °C[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Indophenol is an organic compound with the formula OC6H4NC6H4OH. It is deep blue dye that is the product of the Berthelot's reaction, a common test for ammonia.[2] The indophenol group, with various substituents in place of OH and various ring substitutions, is found in many dyes used in hair coloring and textiles.[3]
Indophenol is used in hair dyes, lubricants, redox materials, liquid crystal displays, fuel cells and chemical-mechanical polishing. It is an environmental pollutant and is toxic to fish.[1][4]
- ^ a b c d Sabnis, R. W. (2007). Handbook of Acid-Base Indicators. CRC Press. p. 196. ISBN 978-0-8493-8219-2.
- ^ Patton, Charles J.; Crouch, S. R. (1977). "Spectrophotometric and kinetics investigation of the Berthelot reaction for the determination of ammonia". Analytical Chemistry. 49 (3): 464–469. doi:10.1021/ac50011a034.
- ^ Horst Berneth (2002). "Azine Dyes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_213.pub2. ISBN 978-3527306732.
- ^ "Indophenol I5763". 500-85-6. Retrieved 2020-06-11.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search